TETRAETHYLAMMONIUM FLUORIDE HYDRATE
Tetraethylammonium fluoride hydrate
CAS: 98330-04-2
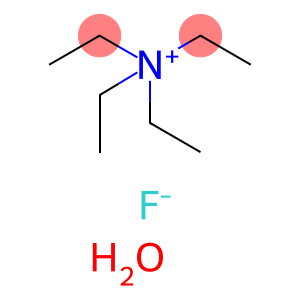
Molecular Formula: C8H22FNO
TETRAETHYLAMMONIUM FLUORIDE HYDRATE - Names and Identifiers
| Name | Tetraethylammonium fluoride hydrate |
| Synonyms | Tetraethylammonium fluoride hydrate TETRAETHYLAMMONIUM FLUORIDE HYDRATE Tetramethylammonium fluoride hydrate Ethanaminium, N,N,N-triethyl-, fluoride, hydrate |
| CAS | 98330-04-2 |
| EINECS | 678-990-4 |
| InChI | InChI=1/C8H20N.FH.H2O/c1-5-9(6-2,7-3)8-4;;/h5-8H2,1-4H3;1H;1H2/q+1;;/p-1 |
TETRAETHYLAMMONIUM FLUORIDE HYDRATE - Physico-chemical Properties
| Molecular Formula | C8H22FNO |
| Molar Mass | 167.26 |
| Water Solubility | Soluble in water. |
| Appearance | Adhering Crystals or Crystals or Crystalline Solid |
| Color | White to beige |
| BRN | 4152659 |
| Storage Condition | Inert atmosphere,Room Temperature |
| Sensitive | Hygroscopic |
| MDL | MFCD00149990 |
TETRAETHYLAMMONIUM FLUORIDE HYDRATE - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S22 - Do not breathe dust. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S28 - After contact with skin, wash immediately with plenty of soap-suds. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| TSCA | Yes |
| HS Code | 29239000 |
| Hazard Note | Irritant |
TETRAETHYLAMMONIUM FLUORIDE HYDRATE - Introduction
Tetraethylammonium fluoride hydrate is an organic salt with the chemical formula (C2H5)4NF · xH2O, where x is the number of water molecules in the crystal. It is colorless crystals, soluble in water and organic solvents.
Tetraethylammonium fluoride hydrate are often used as strong bases in organic synthesis and have corresponding strong alkalinity. It can react with acids to form the corresponding fluoride salts. The compound is used as a base catalyst, a solvent, an ionic liquid component, or the like in organic synthesis. It can also be used to prepare organometallic compounds as catalysts or solvents for non-aqueous systems.
Preparation of Tetraethylammonium fluoride hydrate is usually obtained by reacting tetraethyl ammonia bromide with sodium fluoride. Tetraethylammonium bromide is first reacted with hydrofluoric acid hydrate to give an indirect precursor of tetraethylammonium fluoride. After crystallization and drying, the Tetraethylammonium fluoride hydrate is obtained.
When using Tetraethylammonium fluoride hydrate, appropriate safety precautions should be taken. It is a strong alkali, and contact with skin and eyes may cause burns. Therefore, protective equipment such as gloves and goggles should be worn during operation. In addition, avoid inhaling its dust or solution and avoid prolonged exposure to the substance. If accidentally ingestion, should immediately gastric lavage and medical treatment. During storage, it should be kept in a dry, cool and ventilated place, separated from other substances.
Tetraethylammonium fluoride hydrate are often used as strong bases in organic synthesis and have corresponding strong alkalinity. It can react with acids to form the corresponding fluoride salts. The compound is used as a base catalyst, a solvent, an ionic liquid component, or the like in organic synthesis. It can also be used to prepare organometallic compounds as catalysts or solvents for non-aqueous systems.
Preparation of Tetraethylammonium fluoride hydrate is usually obtained by reacting tetraethyl ammonia bromide with sodium fluoride. Tetraethylammonium bromide is first reacted with hydrofluoric acid hydrate to give an indirect precursor of tetraethylammonium fluoride. After crystallization and drying, the Tetraethylammonium fluoride hydrate is obtained.
When using Tetraethylammonium fluoride hydrate, appropriate safety precautions should be taken. It is a strong alkali, and contact with skin and eyes may cause burns. Therefore, protective equipment such as gloves and goggles should be worn during operation. In addition, avoid inhaling its dust or solution and avoid prolonged exposure to the substance. If accidentally ingestion, should immediately gastric lavage and medical treatment. During storage, it should be kept in a dry, cool and ventilated place, separated from other substances.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: Tetraethylammonium fluoride hydrate Request for quotation
CAS: 98330-04-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 98330-04-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: Tetraethylammonium fluoride hydrate Visit Supplier Webpage Request for quotationCAS: 98330-04-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TETRAETHYLAMMONIUM FLUORIDE HYDRATE Request for quotation
CAS: 98330-04-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 98330-04-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: Tetraethylammonium Fluoride Hydrate Visit Supplier Webpage Request for quotation
CAS: 98330-04-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 98330-04-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Tetraethylammonium fluoride hydrate Request for quotation
CAS: 98330-04-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 98330-04-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: Tetraethylammonium fluoride hydrate Visit Supplier Webpage Request for quotationCAS: 98330-04-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TETRAETHYLAMMONIUM FLUORIDE HYDRATE Request for quotation
CAS: 98330-04-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 98330-04-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: Tetraethylammonium Fluoride Hydrate Visit Supplier Webpage Request for quotation
CAS: 98330-04-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 98330-04-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



